Gene-editing technology has rapidly moved from theoretical science to real-world medical research. If you live with or have heard about rare inherited skin conditions, you may already know how limited treatment options can be. In 2026, gene editing is being explored as a possible way to address the root genetic causes of these disorders rather than just managing symptoms.
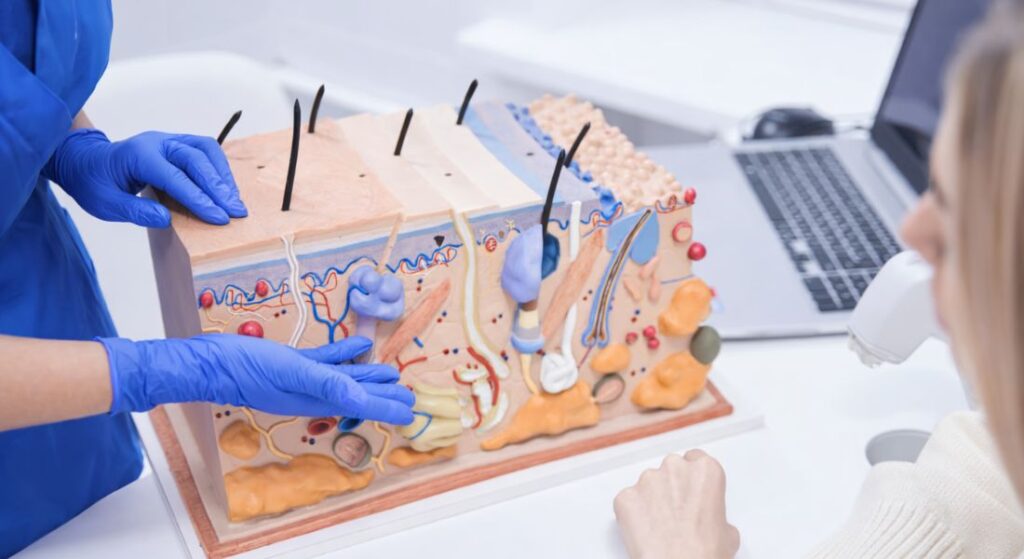
Dermatology is uniquely positioned within gene therapy research because the skin is accessible and visible. Unlike internal organs, skin tissue can be sampled, treated, and monitored directly. This makes it an attractive field for investigating technologies such as CRISPR.
In this article, we examine how gene editing works, what current research is showing, and what realistic expectations look like in 2026. We also explore safety, ethical concerns, and long-term implications. Most importantly, we explain what this evolving science could mean for patients.
What Is Gene Editing?
Gene editing refers to a group of techniques that allow scientists to modify DNA sequences within living cells. In simple terms, it’s about changing the genetic instructions that shape how your body functions. These modifications can remove, repair, or replace faulty genes that may be responsible for disease. The goal isn’t just to manage symptoms you experience, but to correct the underlying cause itself. It’s a very different way of thinking about treatment.
One of the most widely studied tools in this area is CRISPR, which acts a bit like molecular scissors guided by a highly precise targeting system. It allows scientists to cut specific DNA sequences with remarkable accuracy. Unlike traditional therapies that focus on controlling symptoms, gene editing aims to fix the error at its source. Instead of working around the problem, it addresses the genetic blueprint directly. For you, that represents a fundamental shift in how certain diseases might be treated in the future.
Why Dermatology Is a Strong Candidate for Gene Editing
Dermatology is considered a strong candidate for gene editing largely because your skin is one of the most accessible organs in the body. Unlike internal organs, it can be examined directly and monitored over time. Biopsies can be performed relatively safely and, if needed, repeated. That makes it easier for researchers to study how genetic corrections behave in the long term. You’re not relying solely on indirect measurements.
Another advantage is that your skin cells can be grown in laboratory settings. Scientists are able to edit these cells outside your body before placing them back in a controlled way. This approach allows careful testing before any reintroduction takes place. It adds an extra layer of safety and precision. For you, that means potential treatments can be evaluated thoroughly before clinical use.
Because changes in the skin are visible, outcomes can be monitored directly. Improvements in skin strength, healing, or overall integrity can be observed clinically. You and your clinician can often see progress rather than relying only on scans or blood tests. This provides clear, measurable endpoints for research trials. It’s one reason dermatology is seen as such a promising field for gene-editing therapies.
Understanding CRISPR Technology

CRISPR stands for Clustered Regularly Interspaced Short Palindromic Repeats, which might sound complex at first. It was originally discovered as part of a natural immune defence system in bacteria. Scientists later realised this system could be adapted into a powerful gene-editing tool. In simple terms, it gives researchers a way to target very specific sections of DNA. For you, that means potential treatments can be designed with remarkable accuracy.
CRISPR works by guiding a protein called Cas9 to a precise DNA sequence within a cell. You can think of Cas9 as a pair of molecular scissors. Once it reaches the target, it cuts the DNA strand at that exact point. After the cut is made, the cell’s own repair machinery steps in to fix the break. This natural repair process is what makes editing possible.
Researchers can carefully influence how the DNA is repaired after it’s cut. This allows them to delete faulty genes or correct specific genetic errors. The real strength of CRISPR lies in its precision and efficiency. Instead of making broad, unpredictable changes, it focuses on clearly defined targets. For you, that level of control represents a significant step forward in genetic medicine.
Inherited Skin Disorders as Targets
Many inherited skin disorders are caused by mutations in a single gene. If you’re affected by one of these conditions, it often means the problem can be traced back to a specific genetic error. Conditions such as epidermolysis bullosa are well-known examples. They can lead to severe blistering, fragile skin, and chronic wounds that significantly affect daily life. For you, that can mean ongoing care and constant management.
At present, most treatments focus on wound care and controlling symptoms. They aim to protect your skin, reduce infection risk, and improve comfort. However, they don’t correct the underlying genetic mutation that causes the condition in the first place. That’s where gene editing represents a potential shift. Instead of managing the consequences, it seeks to address the root cause.
By repairing the defective gene itself, there is the possibility of long-term disease modification. In theory, you wouldn’t just be treating symptoms, but altering the course of the condition. Research is still ongoing to determine how feasible and safe this approach is. Early findings are cautiously promising, but careful evaluation continues. For you, this area of research offers hope, balanced with scientific realism.
Epidermolysis Bullosa and CRISPR
If you’re looking at where gene-editing research is making real progress, Epidermolysis Bullosa is one of the most closely studied conditions. It’s caused by inherited mutations that affect the structural proteins responsible for keeping your skin layers firmly attached. When those proteins are faulty, even minor friction can cause painful blistering and skin fragility.
Why gene editing makes sense here: Because the condition results from a specific genetic mutation, it’s a logical target for technologies such as CRISPR. If the faulty gene can be corrected in your skin cells, the underlying structural weakness may be improved rather than simply managed.
Encouraging laboratory findings: In laboratory studies, researchers have successfully corrected mutations in affected skin cells. These edited cells demonstrate stronger structural stability and improved protein production. That suggests genuine therapeutic potential at a biological level.
Challenges in clinical translation: Moving from laboratory success to real-world treatment is complex. Safety must be thoroughly validated. Researchers need to ensure gene correction is precise, durable, and free from unintended effects. Long-term stability is particularly important in genetic therapies.
Measured but steady progress: Development continues cautiously and methodically. While gene editing is not yet routine clinical care, each stage of research brings greater clarity and refinement.
For you, the significance lies in the shift in thinking. Instead of only managing symptoms, gene-editing research aims to address the root genetic cause. It’s not an immediate solution, but it represents meaningful scientific momentum in the right direction.
Other Genetic Skin Conditions Under Investigation
Beyond epidermolysis bullosa, researchers are also exploring other inherited skin conditions. These include disorders such as ichthyosis and certain pigmentary conditions that arise from specific gene mutations. If you’re living with one of these, the cause is often clearly defined at a genetic level. That clarity makes them potential candidates for targeted gene-editing approaches. The more precise the mutation, the more focused the strategy can be.
When scientists can identify a well-defined genetic fault, it becomes easier to design a tailored intervention. You’re not dealing with a vague or multifactorial cause, but a specific error in the DNA sequence. That improves the feasibility of correction and supports more precise therapeutic planning. In theory, this level of accuracy could lead to more meaningful long-term results. Precision at the genetic level strengthens overall treatment design.
However, much of this research is still experimental. Human trials remain limited, although they are gradually expanding as safety data grows. You won’t see widespread clinical use overnight. Ethical oversight is strict, and rightly so, given the implications of altering genetic material. Progress is steady, but always guided by caution and rigorous evaluation.
Ex Vivo vs In Vivo Gene Editing
When you hear about gene editing, it’s important to understand that there are different approaches. Ex vivo editing involves modifying cells outside your body. In dermatology, that usually means extracting some of your skin cells, editing them in a laboratory, and then reintroducing them.
Because the editing happens outside the body, researchers can test and verify the results beforehand. They can check whether the genetic correction worked as intended and screen for potential unintended effects. Only once the cells meet strict safety criteria are they reintroduced. This staged approach helps reduce risk.
In contrast, in vivo editing involves delivering the gene-editing tools directly into your body. Instead of modifying cells in the lab, the correction takes place within your tissues. This approach is more complex and technically demanding. Precision targeting becomes absolutely essential to avoid affecting unintended cells.
At present, most dermatological research focuses on ex vivo techniques. Controlled editing outside the body helps minimise unintended consequences. It allows for careful oversight before treatment is completed. While in vivo approaches are being explored, safety remains the guiding principle.
Delivery Systems in Gene Editing
For gene editing to work effectively, the editing tools need a reliable way to enter your cells. Viral vectors are commonly used for this purpose, as they’re naturally good at delivering genetic material. Scientists modify these viruses so they can transport CRISPR components safely. At the same time, non-viral systems are also being explored to reduce potential risks. For you, the method of delivery is just as important as the edit itself.
Nanotechnology may play an increasingly important role in future delivery systems. By combining gene editing with advanced carriers, researchers hope to enhance precision and control. This interdisciplinary approach brings together genetics, molecular biology, and materials science. The efficiency of delivery often determines whether a therapy succeeds or fails. That’s why ongoing research continues to refine safer and more targeted systems designed with your wellbeing in mind.
Safety Considerations
Gene editing is a powerful tool, but it does carry inherent risks. One of the main concerns is something known as off-target effects, where unintended regions of your DNA are altered. Even small, unintended changes could have consequences. That’s why minimising these errors is a major research priority. Precision isn’t just desirable it’s essential for your safety.
Long-term monitoring is equally important. Even if an edit appears accurate at first, researchers need to evaluate how durable it is over time. You wouldn’t want a correction that fades or creates unexpected effects years later. Ongoing follow-up helps identify any unintended consequences early. Patient safety remains at the centre of every stage of development.
Strict regulatory frameworks guide this research from start to finish. Clinical trials undergo extensive scientific and ethical review before they begin. You’re not simply relying on laboratory success; you’re relying on structured oversight. Transparency in reporting results is vital for maintaining trust. Every advance is carefully measured to ensure progress never outpaces safety.
Ethical Concerns
Gene editing doesn’t just raise scientific questions; it raises ethical ones too. Altering DNA is a serious step, and you’d expect long-term implications to be considered carefully. Changes at the genetic level can carry consequences that extend beyond immediate results. Public trust depends on research being conducted responsibly and transparently. Without that trust, progress would quickly lose credibility.
It’s also important to understand the difference between somatic and germline editing. Somatic editing affects only you, the treated individual, and does not pass changes on to future generations. Germline editing, however, would alter DNA in a way that could be inherited, which makes it far more controversial. In dermatology, research is focused on somatic correction rather than germline modification. That distinction plays a key role in ethical discussions.
Ethical oversight committees monitor studies closely to ensure standards are upheld. Informed consent is essential, so you fully understand potential risks and benefits before participating in any trial. Transparency in methods and results helps maintain accountability. Responsible innovation remains the guiding principle in this field. Progress is encouraged, but never at the expense of ethical integrity.
Durability of Gene Correction
If you’re considering the promise of gene editing, one of the most important questions is simple: will it last? Correcting a mutation is only truly meaningful if those edited cells survive, integrate properly, and continue functioning long-term. Durability ultimately determines whether a therapy is transformative or temporary.
Integration and survival of edited cells: When skin cells are genetically corrected, they must not only produce the right protein they also need to integrate into your existing tissue architecture. If those cells fail to persist, the therapeutic effect may fade.
The role of stem cells: Your skin is constantly regenerating. That means corrected surface cells alone aren’t enough. Ideally, gene editing needs to occur in epidermal stem cells, so that as your skin renews itself, the corrected genetic instructions are passed on. Sustained benefit depends on these long-lived cells maintaining function over time.
Ongoing evaluation of stability: Researchers are closely studying whether corrected cells remain stable, continue expressing healthy protein levels, and avoid unintended genetic changes. Monitoring is meticulous, particularly in early clinical trials.
Long-term data is still emerging: At present, long-term follow-up data is limited. Early trial participants continue to be observed to assess persistence of benefit and safety. Only time will confirm whether gene correction provides durable, lifelong improvement.
For you, durability is the defining factor. Gene editing holds enormous potential but its real value lies not just in correcting DNA, but in ensuring that correction endures as your skin renews and evolves.
Potential Benefits for Patients
Gene editing offers real hope when it comes to disease modification. If a faulty gene can be corrected, you may no longer experience the same recurring symptoms. Rather than managing flare-ups again and again, the goal would be to address the root cause. For you, that could mean a meaningful improvement in quality of life. It has the potential to change how certain inherited skin conditions are experienced altogether.
Another possible benefit is reduced reliance on ongoing treatment. Instead of lifelong symptom control, you might see more lasting improvement after a targeted intervention. That would represent a genuine shift in how chronic genetic conditions are managed. However, expectations need to remain realistic, as research is still developing. Widespread clinical application isn’t immediate, but progress continues steadily and responsibly.
Regulatory Landscape in 2026
In 2026, regulatory authorities continue to maintain strict control over gene-editing trials. Before any treatment can move forward, extensive safety data must be reviewed. You wouldn’t expect such powerful technology to advance without careful scrutiny. Patient protection remains the top priority at every stage. Approval processes are deliberately thorough rather than rushed.
International guidelines also play an important role in ensuring ethical compliance. Research centres often collaborate across borders to promote consistency and shared standards. That collaboration helps prevent gaps in oversight and strengthens accountability. For you, it means the science is being evaluated within a global framework. Robust supervision remains central to progress.
By 2026, some early trials have moved forward cautiously, showing measured progress. However, widespread approval for routine clinical use is still pending. Evaluation continues as long-term safety and effectiveness data are gathered. You’re seeing advancement, but not acceleration at the expense of safety. Careful assessment remains the guiding principle.
Limitations of Current Research
Gene editing may sound revolutionary, but it’s technically complex. Not every mutation can be corrected easily or safely. Some skin conditions involve multiple genes rather than a single, clearly defined fault. If that’s the case, designing an effective edit becomes far more challenging. For you, that means progress can vary depending on the specific condition.
Even when a mutation is well understood, translating that knowledge into a reliable treatment isn’t straightforward. The editing process must be precise, controlled, and carefully validated. Small errors at the genetic level can have significant consequences. That’s why researchers move methodically rather than quickly. Precision always takes priority over speed.
Cost and scalability are also important barriers. The laboratory techniques involved require highly specialised infrastructure and trained expertise. You’re not just talking about a simple prescription you’re talking about advanced biomedical engineering. Initially, access may be limited to specialised research centres. Broader availability would take time to develop.
Although the research is promising, it’s still in relatively early stages. Clinical application requires further validation through carefully designed trials. You won’t see overnight transformation in routine practice. Caution remains entirely appropriate given the stakes involved. Progress is steady, but it is deliberately measured.
Long-Term Monitoring Requirements
If you take part in a gene-editing trial, ongoing follow-up would be essential. Monitoring isn’t just about immediate results; it’s about ensuring long-term safety and durability. Researchers need to understand how the genetic changes behave over time. Regular assessments help confirm that the correction remains stable. For you, that means continued medical oversight rather than a one-off intervention.
Long-term registries are often established to track outcomes across groups of participants. These databases allow researchers to identify patterns and detect any adverse effects as early as possible. If unexpected issues arise, they can be investigated quickly. Transparency in reporting findings plays a crucial role in maintaining public trust. You benefit from a system designed to prioritise openness and accountability.
Comprehensive monitoring frameworks are therefore a central part of responsible gene-editing research. Patient welfare remains the guiding principle at every stage. Oversight doesn’t end once a procedure is completed. Continued evaluation ensures that progress remains evidence-based and safe. In the long run, that careful supervision protects both you and the integrity of the science.
Combining Gene Editing With Other Therapies
Gene editing doesn’t necessarily replace existing treatments; in many cases, it may work alongside them. You might see combined approaches designed to enhance overall results rather than relying on a single intervention. These multimodal strategies are currently being explored in research settings. The idea is to use each therapy for what it does best. For you, that could mean a more balanced and effective treatment plan.
Even if gene editing proves successful, supportive wound care or conventional management may still be needed at first. Your skin may require time to adjust as corrected cells gradually integrate. Recovery and stabilisation don’t happen instantly. A hybrid approach could help bridge that transition period safely. Over time, such combinations may become more common in clinical practice.
Researchers are continuing to evaluate which combinations deliver the best outcomes. Synergy between therapies may strengthen effectiveness while maintaining safety. You’re likely to see innovation focused on integration rather than replacement. Progress in this field remains dynamic and evidence-driven. Ultimately, the goal is to design strategies that work cohesively for your long-term benefit.
Public Perception and Education
Public understanding of gene editing can vary quite widely. You might come across headlines that make dramatic claims about breakthroughs or cures. Media coverage sometimes oversimplifies complex scientific progress. While innovation is real, it’s rarely as immediate as it sounds. Clear, balanced communication is essential so you’re not misled.
As a patient, you deserve realistic and evidence-based information. Hype should never replace carefully gathered scientific data. When expectations are inflated, disappointment and mistrust can follow. Honest discussion about benefits, risks, and limitations is far more valuable. Education is what allows you to form informed opinions.
Healthcare professionals play a crucial role in guiding these conversations. They help translate complex research into language you can understand. Accurate explanations allow you to weigh potential options thoughtfully. Without that clarity, decision-making becomes far more difficult. Informed choice depends on accessible information.
Transparency ultimately strengthens public confidence. When researchers and clinicians openly share both successes and limitations, trust grows naturally. You’re more likely to feel comfortable with new technologies when you understand how they work and how they’re regulated. Responsible communication supports responsible progress. In the end, knowledge empowers you to engage confidently with emerging therapies.
Future Directions
Research momentum is continuing into 2026 and beyond. You’re seeing more refined precision tools being developed, designed to reduce off-target effects and improve reliability. Editing efficiency is gradually increasing, which means interventions may become safer and more consistent over time. Progress isn’t dramatic overnight it’s careful, step-by-step refinement built on solid data.
New delivery systems may also expand what’s possible. As targeting improves, a broader range of dermatological conditions could realistically become candidates for gene editing. Innovation in this space remains rapid, but measured. The field is evolving steadily rather than suddenly, and that’s actually reassuring meaningful breakthroughs take time, and patience remains essential.
What This Means for Patients Today

Gene editing isn’t yet part of routine clinical practice in dermatology. At present, it remains largely within research and trial settings. For you, that means access is limited to specialised studies rather than standard treatments. Still, the progress being made is tangible and gives a clear sense of where the field is heading. Early results show promise, even if widespread availability is some way off.
If you live with an inherited skin disorder, staying informed can be valuable. Clinical trials may expand over the next few years, potentially giving you access to novel therapies. Speaking with dermatology specialists helps provide clarity about what’s available and what may be realistic for your condition. Knowledge helps you plan and make informed decisions about your care.
Managing expectations is key. While the science is promising, gene editing is still developing, and immediate results shouldn’t be assumed. Responsible optimism is the best approach you can be hopeful, but it’s important to understand the current limitations. The field is progressing carefully, with your safety always at the centre.
FAQs:
1. What is gene editing and how could it help my skin condition?
Gene editing allows scientists to change your DNA to fix faulty genes causing disease. For inherited skin disorders, it targets the root cause rather than just managing symptoms. This could potentially alter how your condition progresses and improve your quality of life.
2. Why is dermatology ideal for gene-editing research?
Your skin is easy to access and monitor, which makes testing safer and more precise. Researchers can edit cells in the lab and then reintroduce them while tracking results directly. Visible changes also allow you and your doctor to see progress clearly.
3. How does CRISPR work?
CRISPR uses a protein called Cas9, which acts like molecular scissors to cut specific DNA sequences. Your cells then repair the DNA, allowing faulty genes to be corrected. This precision makes targeted treatments possible without affecting other genes.
4. Which skin conditions are being targeted?
Inherited disorders like epidermolysis bullosa, ichthyosis, and some pigmentary conditions are under study. These conditions are caused by specific gene mutations, making them suitable candidates for gene editing. The goal is to address the genetic cause rather than just symptoms.
5. What’s the difference between ex vivo and in vivo editing?
Ex vivo editing modifies your cells outside your body before putting them back, while in vivo happens directly inside your tissues. Most research focuses on ex vivo because it allows careful testing and reduces risks, keeping safety as a priority.
6. Are there safety risks?
Yes, there’s a risk of off-target DNA changes, which could have consequences. Long-term monitoring and strict regulatory oversight are in place to keep you safe. Researchers take precision very seriously to minimise potential issues.
7. What about ethical concerns?
Current research focuses on somatic editing, affecting only you, not future generations. Ethical oversight ensures informed consent, transparency, and responsible use of this technology. Germline editing, which would be inherited, is not pursued in dermatology.
8. How long do gene-editing benefits last?
Benefits depend on whether corrected cells survive and integrate into your skin long-term. Editing stem cells is key, as these help your skin renew itself with the corrected gene. Early data is promising, but long-term results are still being studied.
9. Will gene editing replace existing treatments?
Not entirely. Supportive care like wound management may still be needed initially. Gene editing could be combined with conventional therapies to improve outcomes and help your skin adjust safely.
10. When will these therapies be available?
Gene editing is still mostly in clinical trials as of 2026. Access is limited to specialised studies, and widespread use will take time. Staying informed and consulting specialists is the best way to understand your options.
Final Thoughts: Gene Editing and the Future of Dermatology
Gene editing represents a significant step forward in the management of inherited skin disorders. While the technology is still evolving, early research using tools like CRISPR shows promise in targeting the root genetic causes rather than just managing symptoms. Progress in dermatology is careful, evidence-driven, and guided by safety and ethical standards, giving hope for more durable and transformative treatments in the future.
If you’re considering a dermatologist in London, you can get in touch with us at London Dermatology Centre to discuss current research, potential clinical trials, and what these emerging therapies might mean for your skin health. Staying informed and seeking expert guidance remains the best way to navigate this rapidly developing field.
References:
1. Wang, X., Wang, X., Li, Y., A, S., Qiu, B., Bushmalyova, A., He, Z., Wang, W. and Lara‑Sáez, I., 2023. CRISPR‑Cas9‑based non‑viral gene editing therapy for topical treatment of recessive dystrophic epidermolysis bullosa. https://pubmed.ncbi.nlm.nih.gov/38027067/
2. Lu, X. and Jin, H., 2023. A review of CRISPR‑based advances in dermatological diseases. Molecular Diagnosis & Therapy,27(4),pp.445–456. doi:10.1007/s40291‑023‑00642‑5. https://pubmed.ncbi.nlm.nih.gov/37041404/
3. Brooks et al. (2023) Focuses on the challenges and key CRISPR approaches for genodermatoses, which is central to safety and ethical sections of your article. https://www.mdpi.com/1422-0067/24/3/2298
4. Leachman, S.A. and Hickerson, R.P., 2025. CRISPR/Cas in dermatology: A scoping review of candidate diseases for clinical applications. JAAD Reviews, 11, pp.100065. https://www.sciencedirect.com/science/article/pii/S2950198925000637
5. Kulkarni, R.P., Wang, J. and Bodmer, W.F., 2021. Advances in gene therapy and their application to skin diseases: A review. Journal of Dermatological Science, 103(1), pp.2–9. https://www.sciencedirect.com/science/article/abs/pii/S0923181121001109
