Modern dermatology is increasingly shaped by molecular science, and microRNA research represents one of the most intriguing frontiers. If you have ever wondered how tiny biological molecules can influence complex skin diseases, you are already thinking along the right lines. MicroRNAs may be small, but their regulatory impact is substantial.
These short RNA sequences do not code for proteins directly. Instead, they regulate how genes are expressed after transcription. This means they influence which proteins are produced and in what quantity.
In skin disease, this regulatory control becomes highly relevant. Inflammation, pigmentation changes, and even malignancy are influenced by gene expression patterns. Understanding microRNA behaviour may transform how we diagnose and treat dermatological conditions in the future.
What Exactly Is MicroRNA?
MicroRNAs are tiny strands of non-coding RNA, usually made up of around 20–25 nucleotides. They’re small almost microscopic in the grand scheme of your biology yet they have surprisingly powerful effects inside your cells. Even though they don’t create proteins themselves, they still play a crucial regulatory role in how your body functions.
Instead of producing proteins, microRNAs bind to messenger RNA (mRNA). When they attach, they can reduce or block the production of certain proteins. In simple terms, they help fine-tune how your genes are expressed, adjusting output rather than switching genes fully on or off.
You can think of microRNAs as genetic regulators. They don’t write the script of your DNA, but they control the volume at which parts of it are played. That subtle control can have a significant impact on health and disease, influencing everything from inflammation to cancer development.
Why Gene Regulation Matters in Skin
Your skin is constantly renewing itself and responding to environmental stress, whether that’s UV exposure, pollution or minor injury. Gene expression determines how your skin cells divide, repair damage and defend against threats. MicroRNAs play a quiet but important role in coordinating these processes, helping maintain balance at a cellular level.
When gene regulation functions smoothly, your skin can repair efficiently and keep inflammation under control. But when these regulatory systems become disrupted, problems can start to develop. Even small shifts in molecular signalling can influence how your skin behaves over time.
In inflammatory skin diseases, gene regulation may become dysregulated. Overactive pathways can drive persistent inflammation, leading to chronic symptoms. MicroRNAs may either amplify these inflammatory signals or suppress them, depending on which pathways are involved.
Understanding these molecular shifts could help clinicians predict how severe a condition might become. Early changes at the genetic regulation level may appear before visible symptoms worsen. This is where precision medicine becomes particularly powerful using deeper biological insight to guide more personalised and proactive care for you.
MicroRNA and Inflammatory Skin Disease
Conditions such as psoriasis and eczema involve complex immune signalling within your skin. MicroRNAs help regulate cytokine production and influence how immune cells behave. When these signals become unbalanced, they can shape the intensity and persistence of inflammation you experience.
Researchers have identified specific microRNA signatures linked to active disease. These molecular patterns may correlate with flare severity, offering insight into how aggressively a condition is behaving beneath the surface. Although the science is still developing, efforts are ongoing to identify reliable and consistent markers.
If microRNA profiling becomes more accessible in clinical practice, it could significantly improve how your condition is monitored. Treatment decisions might become more personalised, based on your unique molecular profile rather than symptoms alone. That level of precision management has the potential to improve long-term outcomes and reduce unnecessary trial and error.
MicroRNA in Acne and Sebaceous Activity
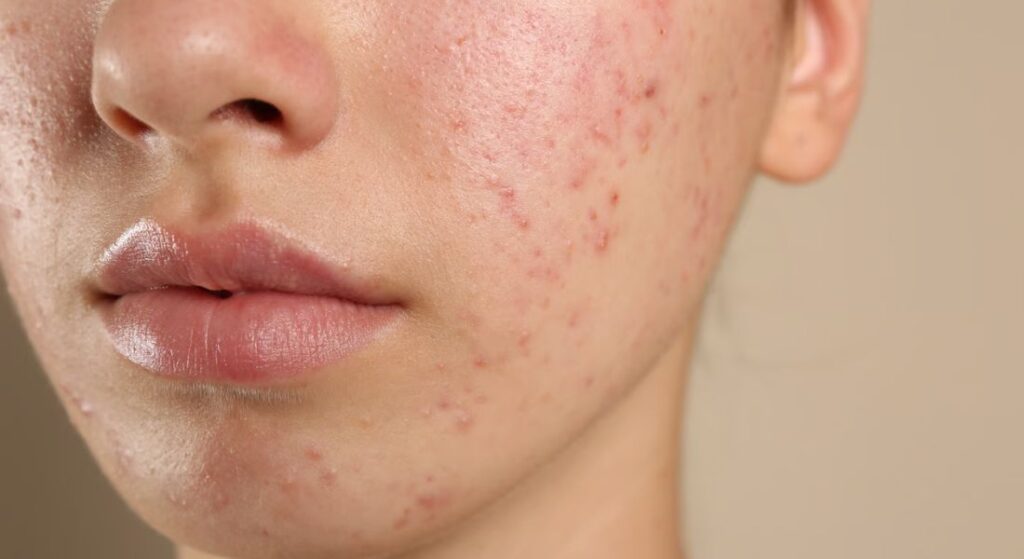
If you struggle with acne, you already know it’s more than just blocked pores. Your skin is dealing with inflammation, bacterial interaction, and overactive sebaceous glands all at once. Researchers are now looking at tiny regulators called microRNAs, which appear to influence how strongly your skin’s inflammatory pathways respond.
1. How microRNAs may affect inflammation: MicroRNAs help control which genes are switched on or off inside your cells. In acne, certain microRNAs seem to regulate inflammatory mediators within your skin. If these signals are amplified, you may experience more redness, swelling, and persistent lesions.
2. Elevated microRNAs in active acne: Studies suggest that specific microRNAs are found at higher levels in active acne lesions. These molecules may contribute to an exaggerated inflammatory response. For you, this could help explain why some breakouts become particularly inflamed or resistant to standard treatment.
3. Potential for targeted therapies: Future treatments may aim to adjust microRNA expression directly. Instead of broadly suppressing inflammation with systemic medication, therapies could fine-tune these molecular regulators at a local level. For you, this might mean more precise control with fewer widespread side effects.
4. Where research stands: This field is still evolving, and clinical applications are under investigation. While the science is promising, large-scale trials are needed before microRNA-targeted treatments become routine practice.
In the end, understanding microRNAs adds another layer to how we view acne not just as a surface issue, but as a carefully regulated biological process. As research progresses, your future acne treatments may become more targeted, personalised, and biologically precise.
Pigmentary Disorders and Gene Expression
Your skin pigmentation depends on how well your melanocytes function and how efficiently melanin is produced. These processes are controlled by complex enzyme pathways and microRNAs help regulate many of them. Because of this influence, scientists are increasingly interested in whether adjusting microRNA activity could one day help manage pigmentary conditions.
In disorders such as vitiligo or melasma, altered microRNA profiles have been observed. Disrupted regulation may affect melanocyte survival or alter melanin production, which can change how evenly your skin is pigmented. Studying these molecular patterns could eventually support more accurate diagnosis or help predict how a condition might progress.
At the moment, therapeutic modulation of microRNAs remains experimental. However, the idea of targeting molecular pathways directly is appealing, as it may allow for more specific treatment. If precision control becomes possible, it could reduce unwanted side effects and move pigment therapy closer to truly personalised care for you.
MicroRNA and Skin Cancer
In melanoma and non-melanoma skin cancers, gene expression changes quite dramatically. MicroRNAs are part of that shift. Some act as tumour suppressors, helping to control abnormal cell growth, while others may promote malignant behaviour if they become overactive or dysregulated. That balance can influence how aggressively a cancer behaves in your skin.
Because these patterns change during cancer development, researchers are studying whether microRNA profiling could help detect malignancy earlier. Distinct molecular signatures may one day help differentiate between benign moles and malignant lesions with greater accuracy. For you, that could mean more precise diagnostics alongside traditional examination and biopsy.
There’s also growing interest in whether microRNAs could be used therapeutically. In theory, adjusting specific microRNA levels might slow tumour growth or make cancer cells more responsive to existing treatments. This approach aims to target the molecular drivers rather than just the visible tumour.
That said, clinical translation is still in development. Most of this work remains within research settings, and long-term safety and effectiveness need to be clearly established. The science is promising, but careful evidence will determine how these discoveries ultimately shape future skin cancer care.
Diagnostic Potential of MicroRNA Profiling
MicroRNAs can be detected not only in tissue samples but also in circulating blood, which opens the door to far less invasive diagnostic approaches for you. Instead of relying solely on surgical biopsies, clinicians may one day use molecular signatures to guide treatment decisions more precisely. Profiling these patterns could help identify specific disease subtypes, allowing therapy to be tailored more accurately and improving outcomes through earlier, more targeted intervention.
That said, standardisation is essential before this becomes part of routine care. Researchers need to confirm that results are reproducible and reliable across different laboratories and populations. Clinical implementation demands rigorous validation, because when it comes to your health, promising science must always be backed by solid evidence.
Predicting Treatment Response
One of the most promising applications of microRNA research is predicting how well you might respond to treatment. Certain microRNA profiles may indicate whether you’re likely to benefit from a specific biologic therapy. If that becomes reliable in clinical practice, it could significantly reduce the frustrating trial-and-error approach that often comes with prescribing.
When molecular markers can identify likely responders in advance, treatment becomes more efficient and focused. That means you’re less likely to be exposed to medications that may not work for you. In practical terms, this is what personalised medicine really looks like matching the right treatment to the right patient from the outset.
Research in psoriasis and other inflammatory dermatoses is actively exploring these predictive patterns. As modelling becomes more refined, prescribing strategies may start to incorporate molecular profiling alongside clinical assessment. The goal is simple: improve precision and deliver better, more individualised care for you.
Therapeutic Targeting of MicroRNA
Beyond diagnosis, microRNAs could eventually become direct treatment targets for you. Scientists are exploring whether synthetic molecules can suppress overactive microRNAs that are driving disease. In other cases, if certain protective microRNAs are lacking, they may be replaced or boosted to restore balance within your cells.
At the moment, though, this approach is still experimental. One of the biggest challenges is delivery getting these molecules exactly where they need to go in your skin without them degrading too quickly. Target specificity is crucial, because you wouldn’t want to interfere with healthy pathways while trying to correct a problematic one.
Topical creams or injectable RNA-based therapies are currently under investigation. Before anything reaches routine clinical use, safety, stability and long-term effects must be clearly established. If those hurdles are overcome, controlled microRNA regulation could open the door to highly precise, personalised treatments for you in the future.
Challenges in Clinical Translation

Even though the research around microRNA is promising, bringing it into everyday clinical practice isn’t straightforward. One of the biggest hurdles is delivery. RNA molecules are naturally fragile and can degrade quickly, so any treatment designed for you would need a system that keeps them stable long enough to actually work.
Targeting is another major challenge. Treatments must be precise enough to influence the intended pathway without interfering with other essential processes in your cells. Off-target effects could lead to unintended consequences, which is why safety remains absolutely paramount at every stage of development.
Cost and scalability also come into play. It’s one thing to achieve success in a laboratory setting, and quite another to produce therapies that are affordable and accessible in routine care. For you to benefit widely, manufacturing and distribution need to be practical as well as scientifically sound.
Ultimately, innovation must translate into safe, evidence-based care. Careful trials, regulatory oversight and long-term data are essential before adoption becomes mainstream. That measured approach is what protects patients while allowing genuine progress to move forward.
Ethical and Regulatory Considerations
If you’re hearing about gene-regulating therapies, it’s natural to wonder how closely they’re monitored. Because these treatments influence gene expression, they require strict regulatory oversight. Safety isn’t assumed it’s carefully evaluated at every stage before approval.
1. Why regulation is so rigorous: Regulatory bodies assess laboratory data, clinical trial results, and long-term safety outcomes before allowing wider use. For you, this means multiple layers of review are designed to protect your wellbeing before a therapy ever reaches routine practice.
2. The ethical complexity of gene regulation: Manipulating gene expression raises important questions. What are the long-term consequences? Could unintended pathways be affected? These are not small issues. For you, it’s reassuring to know that caution, not speed, guides innovation in this space.
3. Long-term monitoring is essential: Even after approval, ongoing surveillance continues. Researchers track outcomes to ensure benefits remain consistent and risks stay low. This long-term follow-up strengthens public confidence and scientific integrity.
4. Clear governance builds trust: Transparent research practices and strong ethical frameworks help ensure that innovation serves patients responsibly. When governance is clear, you can feel more confident that new therapies are developed with your safety in mind.
In the end, progress in gene-regulating medicine depends on balance advancing science while maintaining strict ethical standards. Responsible research ensures that potential benefits outweigh risks, keeping patient safety at the centre of every step forward.
Integration with Artificial Intelligence
Artificial intelligence could significantly enhance how microRNA data is analysed for you. Machine learning systems are particularly good at spotting subtle patterns in large, complex molecular datasets patterns that might not be obvious through traditional analysis. By using predictive modelling, clinicians may be able to improve diagnostic accuracy and better anticipate how a condition is likely to behave.
When AI is combined with microRNA profiling, diagnosis and treatment planning could become far more refined. Integrating biological data with algorithmic analysis allows for deeper personalisation, potentially tailoring decisions to your unique molecular signature. As these technologies evolve together, dermatology may become increasingly data-driven, with innovation reshaping how precision care is delivered in everyday practice.
Personalised Dermatology Care

MicroRNA research fits closely with the wider shift towards personalised medicine. Instead of treating skin conditions in a broad, one-size-fits-all way, therapy may increasingly be tailored to you as an individual. With deeper molecular insight, interventions can be guided by what’s actually happening beneath the surface of your skin.
Your unique genetic and molecular profile could one day help inform which treatments are most likely to work for you. That means less unnecessary exposure to medications that may not be effective and a more focused approach from the outset. Precision doesn’t just sound impressive it can genuinely improve effectiveness and efficiency in your care.
Personalised dermatology is widely seen as the future of the field. As molecular markers become better understood, they may increasingly drive clinical decision-making. Over time, this could mean your treatment plan feels far more tailored, grounded in your biology rather than just your visible symptoms.
MicroRNA in Chronic Wound Healing
Chronic wounds often develop because normal gene regulation becomes disrupted. The usual balance between cell growth, inflammation and tissue repair doesn’t function as it should. MicroRNAs play a role in controlling cellular proliferation and inflammatory signalling, so when they’re dysregulated, healing can stall.
Researchers are exploring whether targeting these pathways could help restore balance in your skin. By adjusting specific microRNA expression levels, it may be possible to encourage healthier cell activity and reduce prolonged inflammation. The idea is to nudge the wound environment back towards effective repair rather than chronic stagnation.
Laboratory models have shown promising results, with improved healing rates observed under controlled conditions. However, translating those findings into real-world clinical treatments for you requires careful evaluation. Safety, delivery methods and long-term outcomes all need to be clearly established.
A deeper understanding of the molecular drivers behind chronic wounds is helping shape smarter therapeutic design. Future regenerative strategies may combine established wound care with precise RNA modulation. Research in this area is expanding steadily, offering cautious optimism for more effective long-term solutions.
Role in Autoimmune Skin Conditions
Autoimmune skin conditions develop when your immune system becomes misdirected and starts targeting healthy tissue. MicroRNAs help regulate how immune cells differentiate and function, so when these regulatory signals become disrupted, disease activity can increase. In other words, small molecular imbalances can contribute to much larger inflammatory consequences in your skin.
By identifying abnormal microRNA patterns, clinicians may one day classify autoimmune subtypes more accurately and tailor treatment with greater precision. Biomarker discovery in this area is ongoing, and there’s growing interest in whether therapeutic correction of specific microRNAs could reduce autoimmune activity. Targeted RNA therapies may eventually help modulate immune pathways more selectively but carefully designed clinical trials will determine how feasible and safe that approach truly is for you.
Limitations to Current Evidence
Most microRNA research is still at the preclinical or early clinical stage. While laboratory findings are encouraging, large-scale human trials remain limited. Long-term outcome data is also still emerging, which means conclusions must be drawn carefully rather than prematurely.
What works in controlled laboratory conditions doesn’t always translate directly into real-world clinical practice. Findings need to be validated in diverse patient populations, and reproducibility across different research centres is essential. Translational research moving from bench to bedside inevitably takes time.
It’s natural to feel optimistic about innovation, but enthusiasm needs to be balanced with caution. In medicine, evidence should always guide adoption rather than momentum alone. Ultimately, responsible practice prioritises your safety above all else.
Timeline for Clinical Adoption
If you’re wondering when microRNA-based therapies might become part of routine care, the honest answer is: not immediately. Scientific innovation moves in stages, and each phase requires careful progression. Before anything reaches your clinic, robust safety and efficacy data must accumulate.
1. Therapies will take time: Developing treatments that alter microRNA expression involves laboratory validation, early human trials, and long-term monitoring. For you, this means widespread therapeutic use is still some years away, as regulators require extensive proof of safety.
2. Diagnostics may arrive sooner: Interestingly, diagnostic profiling identifying specific microRNA patterns in your skin may become available before targeted treatments do. Understanding your molecular profile could help guide existing therapies, even before new drugs are approved.
3. Regulatory processes are rigorous: Because gene-regulating approaches influence biological pathways at a fundamental level, approval pathways are understandably strict. For you, this ensures that when a therapy is introduced, it has been thoroughly evaluated.
4. Gradual integration protects patients: Innovation in medicine rarely happens overnight. Step-by-step adoption allows clinicians to gain experience while maintaining safety standards. Patience isn’t a delay it’s part of responsible progress.
In the end, microRNA-based therapies are evolving steadily rather than rapidly. For you, that means today’s research could become tomorrow’s clinical option but only once safety, effectiveness, and regulatory standards are fully satisfied.
Impact on Dermatology Practice
If microRNA profiling is validated in large clinical studies, it could meaningfully refine how your condition is diagnosed and monitored. Dermatologists may begin using molecular data alongside traditional clinical examination, rather than relying on visual assessment alone. For you, this means a more multidimensional evaluation combining what can be seen on the surface with what is happening at a biological level ultimately improving diagnostic precision.
As the science advances, training will inevitably evolve. Future dermatology specialists will need a working understanding of genetic regulation and molecular signalling pathways. For you, that translates into clinicians who are not only skilled in visual diagnosis, but also confident interpreting complex biological data to guide personalised treatment decisions.
Importantly, technology will enhance clinical care, not replace it. Molecular tools may support decision-making, but expert judgement remains central. For you, the integration of advanced profiling with experienced clinical insight has the potential to strengthen overall care quality and improve long-term outcomes.
What This Means for You
Although microRNA-based therapies aren’t part of routine dermatology just yet, the research points towards genuinely transformative potential. In the coming years, you may see more precise diagnostic tools emerging, helping your clinician understand your condition at a deeper biological level. As this evolves, treatment personalisation could expand meaning therapies may be tailored more closely to your individual inflammatory or genetic profile.
Staying informed puts you in a stronger position. When you understand the direction science is moving in, you’re better prepared to discuss new options as they become available. Advances in molecular medicine may gradually reshape how inflammatory and even malignant skin diseases are managed. Progress doesn’t happen overnight, but it does move steadily and those steady steps could significantly influence your future care.
FAQs:
1. What is microRNA and why does it matter in dermatology?
MicroRNA is a tiny strand of non-coding RNA that helps regulate how your genes are expressed. Although it doesn’t create proteins itself, it controls how much of certain proteins are produced. In your skin, that regulation can influence inflammation, pigmentation, healing and even cancer development.
2. How do microRNAs affect skin inflammation?
MicroRNAs help control immune signalling inside your skin cells. If these regulators become unbalanced, inflammatory pathways may become overactive, which can worsen conditions like psoriasis or eczema. Understanding these patterns could eventually help tailor treatment more precisely to you.
3. Could microRNA research improve acne treatment?
Researchers have found that certain microRNAs may influence inflammatory responses in acne. If these signals are amplified, you might experience more severe or persistent breakouts. In the future, therapies could potentially adjust these molecular regulators rather than simply suppressing inflammation broadly.
4. What role do microRNAs play in pigmentary disorders?
Your skin colour depends on well-regulated melanin production, and microRNAs help control those pathways. In conditions like melasma or vitiligo, altered microRNA profiles have been observed. Studying these changes may eventually support more accurate diagnosis and personalised pigment treatments for you.
5. Can microRNAs help detect skin cancer earlier?
MicroRNA patterns often shift when cells become malignant. Researchers are investigating whether specific molecular signatures could help distinguish benign lesions from melanoma more accurately. If validated, this could strengthen diagnostic precision alongside traditional examination and biopsy.
6. Could microRNA profiling predict how you respond to treatment?
One of the most promising areas of research is predicting treatment response. Certain microRNA patterns may indicate whether you’re likely to benefit from specific biologic therapies. This could reduce trial-and-error prescribing and make your treatment journey more efficient.
7. Are microRNA-based treatments available now?
At present, most microRNA therapies remain experimental. While laboratory studies are encouraging, large-scale human trials are still ongoing. Before anything becomes routine in your clinic, long-term safety and effectiveness must be clearly proven.
8. How might microRNAs improve chronic wound healing?
Chronic wounds often involve disrupted gene regulation. MicroRNAs influence cell growth and inflammation, so adjusting their levels may help restore balance in your skin. Early research suggests this could encourage healthier repair, though clinical translation is still developing.
9. Is microRNA testing less invasive than a skin biopsy?
In some cases, microRNAs can be detected in blood samples as well as tissue. This raises the possibility of less invasive diagnostic tools in the future. However, standardisation and validation are essential before such testing becomes part of routine care for you.
10. When could microRNA research change everyday dermatology practice?
You’re unlikely to see widespread clinical adoption immediately. Diagnostics may emerge before targeted therapies do, but full integration into routine dermatology will depend on strong regulatory approval and long-term evidence. Progress is steady, but careful validation always comes first.
Final Thoughts: Small Molecules, Significant Possibilities
MicroRNAs may be tiny, but their potential impact on your skin is anything but small. By influencing how genes are expressed, they offer a deeper layer of insight into why conditions develop, why some treatments work better than others, and how care might become more precise in the years ahead. For you, that could eventually mean fewer unknowns, less trial and error, and treatment decisions guided by your individual biology rather than symptoms alone.
That said, progress in molecular dermatology is steady rather than sudden. Much of the research remains in clinical development, and careful validation is essential before these tools become routine. It’s an exciting direction for science, but responsible medicine always prioritises strong evidence and long-term safety over speed.
If you’re considering seeing a dermatologist in London, contact us at London Dermatology Centre to book a consultation with one of our specialists. Expert assessment ensures your skin concerns are evaluated thoroughly, your options are clearly explained, and your treatment plan reflects both established medical practice and the latest scientific advances.
References:
1. Dopytalska, K., Czaplicka, A., Szymańska, E. and Walecka, I., 2023. The Essential Role of microRNAs in Inflammatory and Autoimmune Skin Diseases A Review. https://www.mdpi.com/1422-0067/24/11/9130
2. Yang, S.C., Alalaiwe, A., Lin, Z.-C., Lin, Y.-C., Aljuffali, I.A. and Fang, J.-Y., 2022. Anti‑Inflammatory microRNAs for Treating Inflammatory Skin Diseases. https://pubmed.ncbi.nlm.nih.gov/36008966/
3. Hawkes, J.E., 2016. microRNAs in Psoriasis. Journal of Dermatological Science, 81, [Review on microRNAs and chronic inflammatory skin disease]. https://www.sciencedirect.com/science/article/pii/S0022202X15000445
4. Singhvi, G., 2018. MicroRNAs as biological regulators in skin disorders. Dermatologic Clinics, Volume [Review on roles of microRNAs in skin pathophysiology]. https://www.sciencedirect.com/science/article/pii/S0753332218341040
5. Jiang, X. et al., 2023. The role of microRNA in psoriasis: A review. Experimental Dermatology, https://pubmed.ncbi.nlm.nih.gov/37382420/
